Services I Services & Capabilities
As a contract research organisation, High Force Research has manufactured chemicals for a wide and diverse range of industries.
Products such as APIs for clinical development, diagnostics, organic electronics, components for medical devices and high- performance materials.
Quality offers consistency and traceability and we have dedicated quality and production units who work in harmony.
Quality encompasses the majority of activities at HFR, some of which might not be immediately obvious. For instance, all personnel, buildings, facilities, documents and records, change control, equipment, materials, analysis, production, packaging, storage and validation are covered by our quality system.
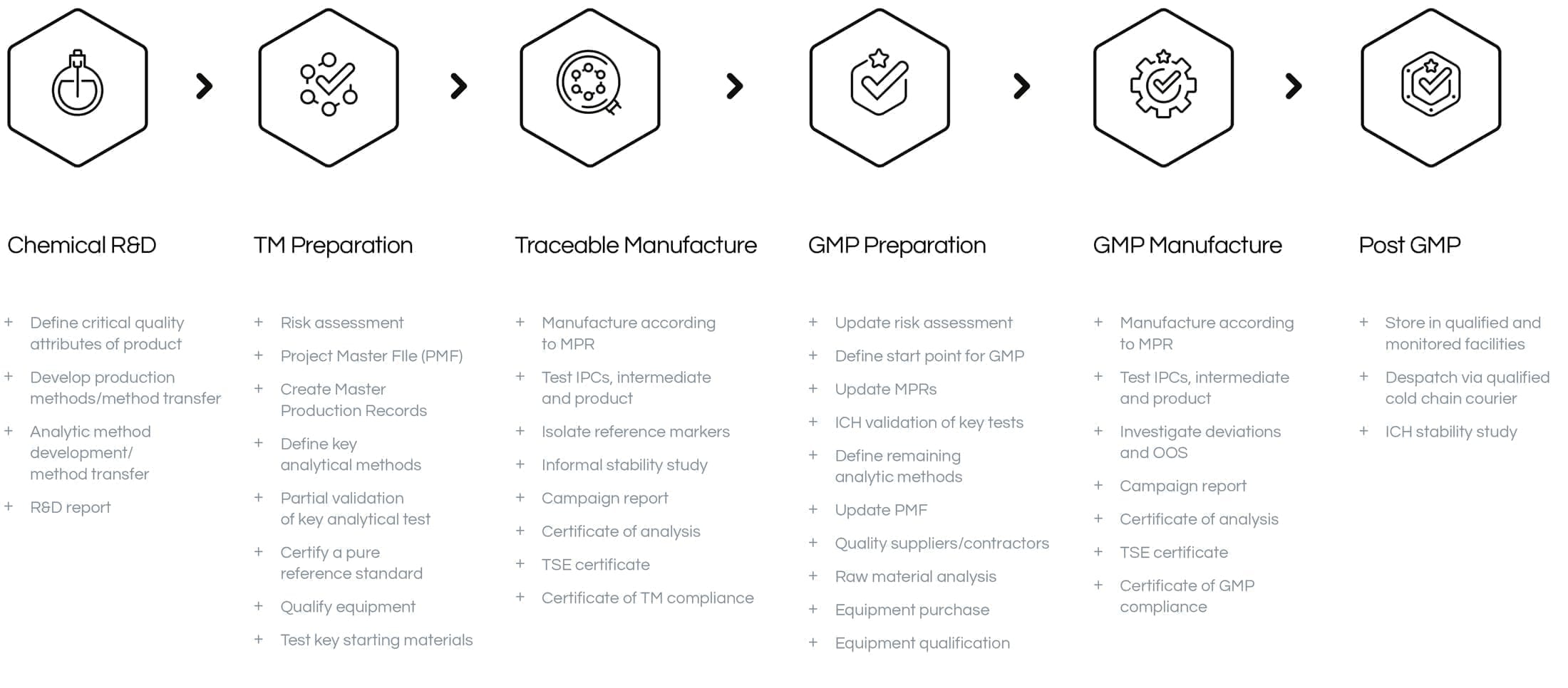
We recently passed an MHRA audit. Continuing our GMP compliance status gives us the ability to manufacture API’s for clinical studies, whilst also allowing us to perform analytical development, validation and stability studies. The highest quality is ensured through our robust in-house auditing.
We also offer traceable manufacture as part of our quality services. This is particularly useful for materials used in pre-clinical studies, API starting materials or performance materials. Traceable manufacture provides consistency which gives our clients the confidence that the materials we produce are the same each and every time, without the stringent controls and extended costs associated with GMP manufacture.

HFR are consistently striving to improve and streamline practices, and have begun preparations to apply for ISO 9001 status.
This internationally recognised standard applies to the whole quality management system. By gaining both accreditations, HFR will demonstrate their ability to deliver high quality and safe products using robust procedures, for different sectors.
In order to increase our value to our clients, we have introduced QPulse, an all-in-one top-class quality management system. This streamlines control and verification of our processes to produce consistent products and services, thereby satisfying the compliance standards of the regulatory bodies.
Understanding and mitigating risk forms a major part of how we build quality into our clients projects.
We think about all the things that can go wrong in a whole process right from your raw materials, how they are going to be supplied, what we need to control – right through to final product and how it will be tested. It becomes a very detailed, carefully thought through process from start to finish, involving different people from different departments within the organisation to feed in to provide something meaningful at the end.
It’s key that our clients are involved during the risk assessment process because quite often we don’t know what critical quality attributes are required for forward manufacture. We have to have those very detailed discussions with the client from the outset, so that we can get a better understanding of what those risks are moving forward and to make sure that we have appropriate controls in place.
Our GMP Journey

Clients and quality
Chemistry can be unpredictable and unfortunately sometimes things do go wrong. We like to keep close contact with our clients, so that if there are any issues we are able to speak to our clients in a very open and honest way. Our clients like to be involved in what actions we take once we come across a problem, so having that relationship is important. It gives our clients confidence in our service and they have input as well.
We help our clients who don’t necessarily have GMP knowledge or experience through the early phases such as what regulatory paperwork needs to be submitted, setting up stability trials and what sort of test methods need to be validated. Often a virtual company won’t have any processes and we’re there to assist them.
HFR has wealth of experience and knowledge of taking projects from a few milligrams up to commercial scale and we are able to provide that support and experience to our clients
Development through to commercialisation requires a hand-in-hand approach involving both chemical innovation and quality. HFR are able to offer support for both, ensuring the smoothest possible path for your project.